Volume: 14 Issue: 1
Prevalence of Diabetic Peripheral Neuropathy among Diabetes Mellitus Patients in a Selected Tertiary Care Center in South India, using Point-of-Care Technology - An Analytical Cross-Sectional study
Year: 2026, Page: 15-21, Doi: https://doi.org/10.47799/pimr.1401.25.82
Received: Nov. 26, 2025 Accepted: Jan. 13, 2026 Published: April 29, 2026
Abstract
Introduction: Diabetic Peripheral Neuropathy (DPN) is a common complication of Type 2 Diabetes Mellitus (T2DM), often underdiagnosed due to limitations of conventional screening tools. Early identification using advanced point-of-care (PoC) technologies is crucial for preventing severe complications. Objectives: To estimate the prevalence of DPN among T2DM patients using a multimodal PoC diagnostic device and examine associations with glycemic control and duration of diabetes. Methodology: A cross-sectional study was conducted among 389 known T2DM patients attending a tertiary care outpatient clinic in South India from February to July 2024. Participants were screened using the Diabetic Neuropathy Symptom (DNS) score and a handheld PoC device assessing touch, vibration, hot, and cold perceptions. Data were analyzed using descriptive statistics and chi-square tests. Results: The mean age of participants was 54.8 ± 10.7 years; 85.3% had uncontrolled glycemic status. Symptom-based DNS scoring identified DPN in 28% of participants. However, 99% were identified at risk using the PoC device, which revealed impairments in both large (vibration, touch) and small (thermal) nerve fibers. Significant associations were found between DPN and both longer diabetes duration (p<0.001) and poor glycemic control (p=0.026). Conclusion: The prevalence of DPN was markedly higher using the PoC device compared to symptom-based methods. This emphasizes the value of integrating multimodal PoC screening into routine diabetic care, especially in rural and resource-limited settings, for early detection and intervention.
Keywords: Diabetic peripheral neuropathy, Point-of-care device, Sensory perception, Glycemic control, Rural healthcar
INTRODUCTION
Diabetic peripheral neuropathy (DPN) is a prevalent and debilitating complication of diabetes mellitus, affecting up to 50% of diabetic patients worldwide. It is characterized by damage to the peripheral nerves, leading to symptoms such as pain, tingling, numbness, and loss of sensation, which significantly impair the quality of life and increase the risk of foot ulcers and amputations [1].
Early detection of DPN is crucial as it allows for timely intervention to prevent further nerve damage and associated complications, such as foot ulcers and amputations [2]. Regular screening is recommended for all diabetic patients, starting at the diagnosis for type 2 diabetes and five years after diagnosis of type 1 diabetes [3].
Various screening tools and devices have been developed to aid in the diagnosis of DPN. Traditional methods include clinical examinations and tests such as the 10-g monofilament test, tuning fork, and nerve conduction studies. However, these methods have limitations in terms of sensitivity, specificity, and the ability to detect early-stage neuropathy [4, 5]. Nerve conduction studies (NCS) are considered the gold standard for diagnosing DPN, but they are not suitable for routine screening due to their high cost, requirement for specialized equipment, and need for trained personnel [6].
In recent years, newer technologies have emerged, including handheld devices that measure multiple sensory modalities such as temperature sensation, vibration, and pressure perception. These devices offer a comprehensive assessment of peripheral nerve function and have shown promise in improving the diagnostic accuracy of DPN screening [7].
Justification
Devices that measure temperature sensation can detect early signs of small fiber neuropathy, which may not be identified by traditional methods [8]. Vibration perception is a key indicator of large fiber function and studies have demonstrated that devices measuring vibration perception provide more consistent and objective results compared to the tuning fork test [9]. Assessing pressure perception with handheld devices can enhance the detection of DPN by providing quantitative measurements that are more reliable than the 10-g monofilament test [10].
Several studies have compared the effectiveness of these newer devices with traditional screening methods. For example, a meta-analysis by Mogilevskaya [9] found that devices measuring multiple modalities had higher sensitivity and specificity for detecting DPN.
Despite progress, further studies are needed to validate these devices’ across settings. Using a point-of-care device in routine tertiary care assessment, we aim to strengthen existing evidence and support earlier detection of DPN.
Objectives
The primary objective of this study is to determine the prevalence of Diabetic Peripheral Neuropathy (DPN) among patients with diabetes mellitus using a point-of-care device.
MATERIALS AND METHODS
This is a Cross-sectional study conducted in a tertiary care center in South India, among the Diabetic patients who attend the MTM-OPD (Non-communicable disease) during February 2024-July 2024. Known cases of Type 2 DM diagnosed at least 1 year prior to the study were included. Type 1 DM and pregnant individuals were excluded. The sample size was calculated using the formula n = Zα2pq/d2, where, Z is the standard normal deviation at the desired confidence level (1.96 for 95% confidence level), p is the estimated prevalence of DPN-39.3% by Darivemula et al., [11] with an absolute precision of 5% and considering a buffer of 10%, the sample size was estimated as 403.
Considering the average monthly attendance as the sampling frame, every 4th OPD patient attending MTM-OPD was included in the study if the inclusion criteria were satisfied. If not, the consecutive individual satisfying inclusion and exclusion criteria were included and proceeded further in the same pattern till the required sample size was achieved.
After informed consent, baseline details were collected using an interviewer-administered Epicollect5 questionnaire. Diabetic Neuropathy Symptom (DNS) Score—a validated four-item tool where any one symptom indicates DPN—was applied, followed by assessment of peripheral neuropathy using a handheld point-of-care device.
Touch, Hot perception, Cold perception, and Vibration sense were examined with the device on both feet at predetermined spots as shown in [Fig. 1]. To assess the vibration sense the vibration probe was gently placed on all predetermined points on the feet and the perceived response from each point was recorded. To assess the touch sensation, the monofilament was pressed against each point at a right angle to the feet gently starting with 10g, followed by 25g and 40 gm, till the touch was perceived and the same was recorded at all the 12 points. Ambient temperature was recorded before initiating the examination for each participant. To assess the cold perception, the Cold probe was gently placed over each point on the feet, and the temperature of the probe was reduced gradually by 1-2oc from the ambient temperature, till the cold sensation was perceived by the study participant. Similarly, to assess the hold perception, the hot probe was gently placed over each point on the feet, and the temperature of the probe was increased gradually by 1-2oC from the ambient temperature, till the hot sensation was perceived by the study participant. Recordings were taken at all 12 points on the feet for both hot and cold sensation perception.
Operational Definition
To quantify the risk for DPN, this study adopted the same criteria on which the hand-held PoC device generates the risk status report, which is given below in [Table. 1].
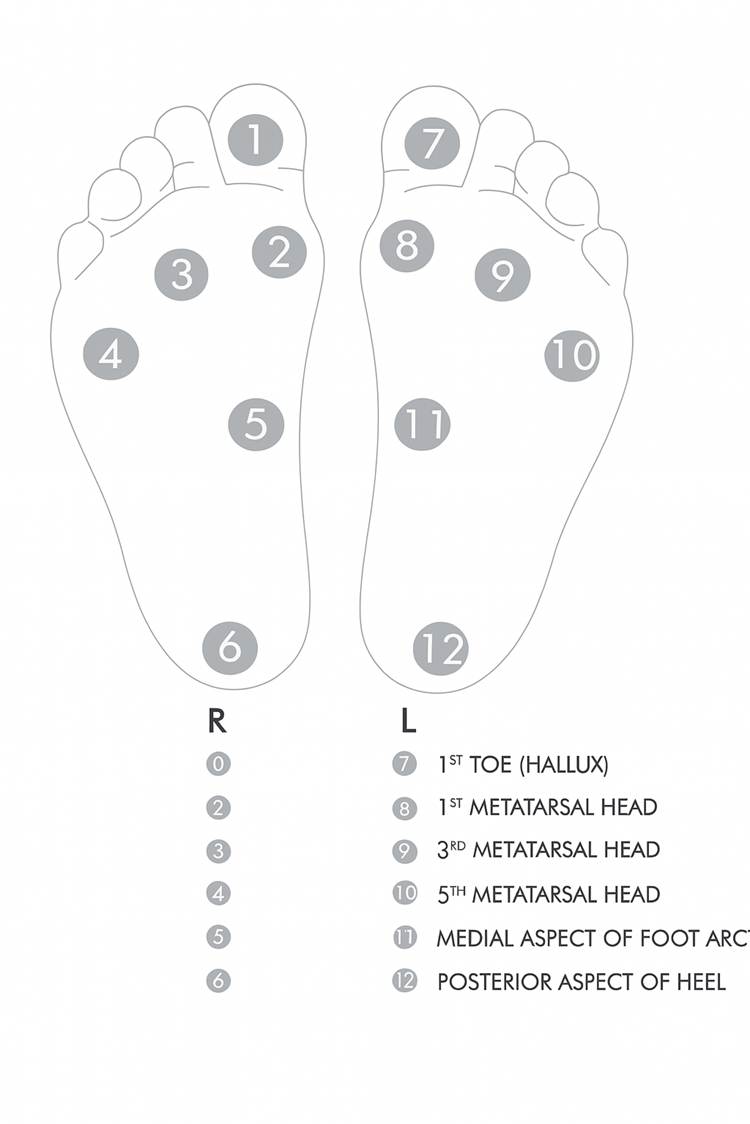
Fig. 1: Points of testing on the feet
| Sensation | Risk category | Criteria |
|---|---|---|
| Touch | Risk - | The tactile sensation felt using 10 grams monofilament at any one of the 4 points - 1st,3rd, and 5th Metatarsal Heads |
| Risk + | Value greater than 10 grams at any one of the 4 points- Hallux,1st,3rd, and 5th Metatarsal Heads | |
| Vibration | Risk - | Vibration detected below 15V |
| Risk + | Vibration detected above 15V | |
| Hot perception | Risk - | A perceived increase in temperature of up to 10°C from the ambient temperature |
| Risk + | Failure to perceive the increase in temperature of up to 10°C from the ambient temperature | |
| Cold perception | Risk - | A perceived decrease in temperature of up to 10°C from the ambient temperature |
| Risk + | Failure to perceive the decrease in temperature of up to 10°C from the ambient temperature |
Table 1: Operation definition to quantify DPN risk
Glycemic Control: Fasting Blood Glucose < 120 mg/dl and PPBS <180 mg/dl in any last recorded value in the past 3 months or Random Blood Glucose < 200 mg/dl. [12]
DPN: All those who fall under Risk for DPN with PoC are considered to be a case of DPN, as no confirmatory test are employed within the scope of this study.
Institutional Ethics Committee approval has been obtained, and all efforts have been made to ensure data privacy and confidentiality at all levels. Services of trained Medical Interns were utilized for collecting data and examination with the hand-held PoC device.
Participants who were identified with risk of DPN have been referred for Neurological consultation and treated as per institutional protocol and the rest were given follow-up appointments after 3 months for routine evaluation. All the study participants were given IEC about Diabetes, its complications, and the importance of Glycemic control.
RESULTS
Out of 400 study recruitments, as 11 participants opted to drop out, the data of 389 participants were taken up for analysis. The mean age of the study participants (n=389) was 54.8 + 10.7 years ranging from 23 to 88 years, most of them belonging to 51-60 years (33.9%). Male and female participants were almost equally represented with 49.1% and 50.9% respectively.
The majority of the study participants were Illiterate (38.6%) and Unemployed (39.1%) with only 4.1% had an education level post-schooling and around 52% belonging to the working class (Skilled /unskilled work). 81.7% (n=318) of the study participants were from rural areas and a majority of them belonged to Socio-economic Class 3 by Modified B.G.Prasad 2024. [Table. 2] shows the demographic characteristics of the study population.
The mean duration of Diabetes Mellitus among the study participants was 5.5 + 4.2 years, with a majority of them belonging to <5 years category with around 7% in the>10 years category.
35.5% (n=138) had associated Systemic Hypertension, and a few proportions had CAD (5.7%), Thyroid disorders (4.4%) and CKD (0.5%), 60.7% (n=236) of the participants had no other associated comorbidities.
89.7% (n=349) of the study participants were on Oral hypoglycemic agents, 3.6% (n=14) were on Insulin and 5.4% (n=21) were both on OHA and Insulin. 5 participants (1.3%) reported that they were not on any treatment.
The mean fasting blood sugar values of the study participants were 165.9 + 54.5 mg/dl (n=147), the mean PPBS was 263.2 + 78.2 mg/dl (n=147), and the mean RBS was 262.4 + 62.1 mg/dl (n=240). 85.3% (n=330) of the participants had uncontrolled glycemic status. Yet, 66.3% (n=258) of participants reported no symptoms of numbness, burning sensation, tingling sensation, or pain in the extremities.
44 study participants (11.3%) reported to have unsteadiness in walking, 83 (21.3%) had Burning or aching pain/tenderness, 62 (15.9%) had pricking sensation and 65 (16.7%) had reported numbness. [Table. 3] shows the DNS scoring scale among the study participants.
| Variable | Category | Frequency | Percentage |
|---|---|---|---|
| Age | <30 years | 7 | 1.8 |
| 31-40 years | 26 | 6.7 | |
| 41-50 years | 118 | 30.3 | |
| 51-60 years | 132 | 33.9 | |
| >60 years | 106 | 27.2 | |
| Gender | Male | 191 | 49.1 |
| Female | 198 | 50.9 | |
| Education | Illiterate | 150 | 38.6 |
| Primary | 82 | 21.1 | |
| Middle | 79 | 20.3 | |
| High | 62 | 15.9 | |
| Diploma | 12 | 3.1 | |
| Graduate | 4 | 1 | |
| Occupation | Unemployed | 152 | 39.1 |
| Unskilled worker | 139 | 35.7 | |
| Semi-skilled worker | 54 | 13.9 | |
| Skilled worker | 10 | 2.6 | |
| Clerical, shop-owner, farmer | 24 | 6.2 | |
| Semi-profession | 2 | 0.5 | |
| Profession | 8 | 2.1 | |
| Residence | Rural | 318 | 81.7 |
| Urban | 71 | 18.3 | |
| SES | Class 1 (Upper) | 83 | 21.3 |
| Class 2 (Upper middle) | 214 | 55 | |
| Class 3 (Middle) | 92 | 23.7 |
Table 2: Demographic Characteristics of the study population
| Diabetic Neuropathy Symptoms |
Frequency | Percentage | |
|---|---|---|---|
| Unsteadiness in walk | Yes | 44 | 11.3 |
| No | 345 | 88.7 | |
| Burning, aching pain / Tenderness |
Yes | 83 | 21.3 |
| No | 306 | 78.7 | |
| Pricking sensation | Yes | 62 | 15.9 |
| No | 327 | 84.1 | |
| Numbness | Yes | 65 | 16.7 |
| No | 324 | 83.3 | |
Table 3: Diabetic Neuropathy Symptoms among the study participants
The proportion of study participants who had PNP as per DNS scale is 28% (n=109). [Table. 4] shows the DPN proportion as per DNS scale.
| DPN status as per DNS score | Frequency | Percentage |
|---|---|---|
| PNP present | 109 | 28.0 |
| PNP absent | 280 | 72.0 |
| Total | 389 | 100.0 |
Table 4: DPN status as per DNS
Duration of Diabetes had a statistically significant association with PNP status as per the DNS scoring scale with a p-value of 0.0001, by Chi-square test statistics. Around 50% of the participants with PNP were in the DM duration category of 5-10 years and about 75% of those without PNP had a duration of DM<5 years.
Similarly, Glycemic control status was also found to be statistically significant with PNP status as per the DNS scoring scale with a p-value of 0.026 by Chi-square test statistics. Around 92% of those with PNP had uncontrolled glycemic status. [Table. 5] shows the association between duration of DM, Glycemic control status and PNP status as per DNS score.
| Variable | Category |
DNS Score |
Total | p-value by Chi-square test | |
|---|---|---|---|---|---|
| PNP Absent | PNP Present | ||||
| Duration of DM | <5 years | 210 | 35 | 245 | <0.0001* |
| 75.0% | 32.1% | 63.0% | |||
| 5-10 years | 62 | 54 | 116 | ||
| 22.1% | 49.5% | 29.8% | |||
| >10 years | 8 | 20 | 28 | ||
| 2.9% | 18.3% | 7.2% | |||
| Glycemic control status | Controlled | 48 | 9 | 57 | <0.026* |
| 17.1% | 8.3% | 14.7% | |||
| Uncontrolled | 232 | 100 | 332 | ||
| 82.9% | 91.7% | 85.3% | |||
Table 5: Association between duration of DM, Glycemic control status and PNP status as per DNS score
The Median value of perception of touch with Microfilament is 12 grams on the right foot and 13.5 grams on the left foot. The Median volts with which the Vibration sense was felt among the study participants were 20.5V and 21.5V on the right and left foot respectively. The Median difference in the temperature of hot perception and the ambient temperature on both feet is 12oC among the study participants. The Median difference in the temperature of cold perception and the ambient temperature is 9oC (Right foot), and 9.5oC (left foot) among the study participants.
The proportion of participants with a risk of DPN using the point-of-care device is 99% (n=385).
DISCUSSION
The mean age of participants in our study was 54.8 ± 10.7 years, with the majority falling in the 51–60-year range (33.9%). This age distribution aligns with findings by Hamasaki et al., who reported DPN more prevalent among individuals aged over 50, reflecting cumulative nerve damage with chronic hyperglycemia [13].
Gender distribution was nearly equal in our study (49.1% males, 50.9% females), and no significant gender-based difference was noted in DPN prevalence. This is consistent with the results from the study by Hideki Kamiya et al., where no substantial sex difference was associated with DPN prevalence using a PoC device [13, 14].
A large proportion of participants were illiterate (38.6%) and unemployed (39.1%), with the majority (81.7%) from rural areas. This socioeconomic profile resembles that of Darivemula et al., who also studied a rural South Indian population and emphasized lower awareness and access to healthcare services as contributory to DPN underdiagnosis [11]. The predominance of lower socioeconomic status may also explain the high proportion of participants (85.3%) with uncontrolled glycemic status, given that resource limitations hinder sustained diabetic care.
The average duration of diabetes was 5.5 ± 4.2 years, with 64% of participants diagnosed within the past five years. A significant association between duration of diabetes and DPN (p < 0.001) was observed, echoing similar findings by Hidetaka Hamasaki et al, who established that longer disease duration increased the risk of impaired sural nerve function [13].
Systemic hypertension was the most common comorbidity (35.5%). Literature indicates that co-existing vascular risk factors such as hypertension exacerbate microvascular complications like DPN by impairing endoneurial blood flow [15].
Only a few participants reported a history of neuropathy-related drug use; most (377 of 389) were not on any such medications. Most commonly reported usage was for amitriptyline (n = 7), followed by pregabalin (n = 3), gabapentin (n = 1), and phenytoin (n = 1).
Only 28% of participants were symptomatic for DPN using the DNS score. Despite this, 99% showed risk of neuropathy on the PoC device. This discrepancy highlights the limitation of symptom-based scales and supports device-assisted early detection. Such asymptomatic detection was emphasized by Kamiya et al., where DPNCheck™ revealed neuropathy in patients with no overt symptoms [14].
The vibration threshold median was 20.5–21.5V, exceeding the diagnostic cutoff (>15V), indicating large fiber involvement. Similarly, touch thresholds and thermal thresholds (cold: ~9°C; hot: ~12°C) suggest small fiber impairment, supporting the dual involvement of nerve fiber types in DPN, consistent with neurophysiological assessments reviewed by Selvarajah et al. [16].
Our study reported a higher prevalence of DPN (99%) using PoC devices compared to 28% by DNS scoring. This sharp contrast mirrors findings by Kamiya et al., where PoC device revealed neuropathy in 60% of asymptomatic patients and Nizar et al., where traditional clinical signs underestimated neuropathy compared to PoC device based assessments [14, 17]. The discordance may be due to small-fiber impairment (detected by thermal thresholds) which often precedes symptoms resulting in early stage DPN [18], cultural/ regional factors as rural patients may underreport pain/numbness due to stoicism or misattribution [19] and device sensitivity as the PoC device’s multimodal approach (vibration, touch, thermal) captures subclinical deficits missed by monofilament tests [20].
In contrast, Darivemula et al. [11] reported a prevalence of 39.3% using clinical tools among rural patients, significantly lower than our device-assisted findings. This discordance reflects the limitation of symptom-only models and reinforces the recommendation by Boulton et al. that objective multimodal testing improves detection of early neuropathic changes [21].
Our study demonstrates a notably higher prevalence of Diabetic Peripheral Neuropathy (DPN), which can be contextually explained by two key factors: Firstly, the study population consisted exclusively of known type 2 diabetic patients with a disease duration exceeding one year, a characteristic associated with increased neuropathic risk. Several studies have established that the duration of diabetes is a strong, independent predictor of DPN, with prevalence increasing substantially after the first year and rising progressively with longer disease durations [13, 22]. Secondly, the study was conducted in a tertiary care referral center, which inherently manages a patient pool with more advanced or complicated diabetic profiles, often referred for specialist care. Such populations are more likely to have poorly controlled glycemia, comorbidities, or pre-existing complications, which contribute to the higher observed prevalence of DPN. This setting-related prevalence inflation has also been noted in studies conducted at specialized centers [11, 14].
Therefore, both the selection of a high-risk cohort (based on disease duration) and the institutional setting (tertiary care referral unit) plausibly explain the elevated DPN rates reported in our study compared to community-based screenings or studies involving recently diagnosed diabetic populations.
The device’s median vibration threshold (20.5–21.5V, exceeding the 15V cutoff) and thermal perception deficits (hot: +12°C; cold: +9°C) suggest concurrent large- and small-fiber damage, consistent with NCS-based studies [23]. While NCS remains the gold standard, PoC devices offer practical advantages for resource-limited settings, as demonstrated by their high detection rates in our study.
CONCLUSION
This study demonstrates a high burden of DPN risk among diabetic patients, with point-of-care multimodal devices showing superior detection capability over symptom-based scores. The prevalence rates observed far exceed those in conventional literature, underscoring the value of objective diagnostic tools, especially in rural, underserved populations. Future research should focus on longitudinal validation and integration of such technologies in routine diabetic care.
Limitations
This study was conducted in a single tertiary care center, which may limit the generalizability of findings to primary care or community settings. The cross-sectional design precludes causal inference between DPN and associated factors. Nerve conduction studies, the gold standard for DPN diagnosis, were not performed for validation of the point-of-care device and the very high “at-risk” proportion detected by the PoC device warrants cautious interpretation and further validation in diverse populations. Possible confounders such as dyslipidemia, smoking and alcohol use, nutritional deficiencies (e.g., vitamin B12), chronic drug history and other causes of peripheral neuropathy were not addressed statistically in this study, which may have influenced the observed associations.
DISCLOSURE
Funding and Conflict of Interest
The authors report no funding or conflicts of interest; the NeuroTouch device was routinely procured with no benefits from its manufacturer.
References
1. Preston FG, Riley DR, Azmi S, Alam U. Painful Diabetic Peripheral Neuropathy: Practical Guidance and Challenges for Clinical Management. Diabetes, Metabolic Syndrome and Obesity. 2023; Volume 16 Available from: https://doi.org/10.2147/dmso.s370050
2. Burgess J, Frank B, Marshall A, Khalil RS, Ponirakis G, Petropoulos IN, et al. Early Detection of Diabetic Peripheral Neuropathy: A Focus on Small Nerve Fibres. Diagnostics. 2021; 11 (2). Available from: https://doi.org/10.3390/diagnostics11020165
3. American Diabetes Association. Classification and diagnosis of diabetes: standards of medical care in diabetes—2021. Diabetes Care. 2021; 44 (Suppl 1). Available from: https://doi.org/10.2337/dc21-s002
4. Carmichael J, Fadavi H, Ishibashi F, Shore AC, Tavakoli M. Advances in Screening, Early Diagnosis and Accurate Staging of Diabetic Neuropathy. Frontiers in Endocrinology. 2021; 12 Available from: https://doi.org/10.3389/fendo.2021.671257
5. Hazari A, Mishra V, Kumar P, Maiya A. The accuracy of 10 g monofilament use for clinical screening of diabetes peripheral neuropathy among Indian population. PLoS One. 2024; 19 (2). Available from: https://doi.org/10.1371/journal.pone.0297110
6. Selvaraj K, Deshmukh P, Phatak M, Patil A, Charlette L, Khot R, et al. Diagnostic Accuracy of Screening Tests for Diabetic Peripheral Neuropathy Among Outpatient Attendees of Follow-Up Care for Diabetes in Central India. Cureus. 2025; 17 (9). Available from: https://doi.org/10.7759/cureus.91706
7. García de la Peña R, Ortiz Romero M, Juárez Jiménez JM, Rayo Pérez AM. Application of NerveCheck Master in the Diagnosis of Diabetic Peripheral Neuropathy. Diabetology. 2025; 6 (1). Available from: https://doi.org/10.3390/diabetology6010003
8. Huang YC, Chuang YC, Chiu WC, Huang CC, Cheng BC, Kuo CEA, et al. Quantitative thermal testing as a screening and follow-up tool for diabetic sensorimotor polyneuropathy in patients with type 2 diabetes and prediabetes. Frontiers in Neuroscience. 2023; 17 Available from: https://doi.org/10.3389/fnins.2023.1115242
9. Mogilevskaya M, Gaviria-Carrillo M, Feliciano-Alfonso JE, Barragan AM, Calderon-Ospina CA, Nava-Mesa MO. Diagnostic Accuracy of Screening Tests for Diabetic Peripheral Neuropathy: An Umbrella Review. Journal of Diabetes Research. 2024; 2024 (1). Available from: https://doi.org/10.1155/jdr/5902036
10. Dube S, Hulke SM, Wakode SL, Khadanga S, Thakare AE, Bharshankar RN, et al. Effectiveness of Semmes Weinstein 10 gm monofilament in diabetic peripheral neuropathy taking nerve conduction and autonomic function study as reference tests. Journal of Family Medicine and Primary Care. 2022; 11 (10). Available from: https://doi.org/10.4103/jfmpc.jfmpc_195_22
11. Darivemula S, Nagoor K, Patan SK, Reddy NB, Deepthi CS, Chittooru CS. Prevalence and its associated determinants of diabetic peripheral neuropathy in individuals with type 2 diabetes mellitus in rural South India. Indian Journal of Community Medicine. 2019 Apr-Jun;44(2):88–91.
12. Government of Tamil Nadu. Makkalai Thedi Maruthuvam – Special Programme for Comprehensive Healthcare. Chennai: Tamil Nadu Health Department; 2022 [cited 2025 Jun 3]. Available from: https://spc.tn.gov.in/makkalai-thedi-maruthuvam/
13. Hamasaki H, Hamasaki Y. Diabetic Neuropathy Evaluated by a Novel Device: Sural Nerve Conduction Is Associated with Glycemic Control and Ankle–Brachial Pressure Index in Japanese Patients with Diabetes. Frontiers in Endocrinology. 2017; 8 Available from: https://doi.org/10.3389/fendo.2017.00203
14. Kamiya H, Shibata Y, Himeno T, Tani H, Nakayama T, Kato M, et al. Point-of-care nerve conduction device predicts the severity of diabetic polyneuropathy: a quantitative but easy-to-use prediction model. Journal of Diabetes Investigation. 2020;11(4):946–952. Available from: https://pubmed.ncbi.nlm.nih.gov/32799422/
15. Said G. Diabetic neuropathy—a review. Nature Clinical Practice Neurology. 2007; 3 (6). Available from: https://doi.org/10.1038/ncpneuro0504
16. Selvarajah D, Kar D, Khunti K, Davies MJ, Scott AR, Walker J, et al. Diabetic peripheral neuropathy: advances in diagnosis and strategies for screening and early intervention. Lancet Diabetes Endocrinol. 2019; 7 (12). Available from: https://doi.org/10.1016/s2213-8587(19)30081-6
17. Nizar H, Munro N, Nightingale P, Fehrer MD. Diagnostic accuracy of the VibraTip in detection of diabetic peripheral neuropathy. British Journal of Diabetes. 2014; 14 (1). Available from: https://doi.org/10.15277/bjdvd.2014.005
18. Al-Khafaji M, Otti V. Early Diagnosis of Diabetic Neuropathy: A Review of Current Diagnostic Tests. Cureus. 2025; 17 (11). Available from: https://doi.org/10.7759/cureus.97880
19. Vyas VA. Pain Management in Indian Older Adults: A Comprehensive Review. Journal of the Indian Academy of Geriatrics. 2025; 21 (2). Available from: https://doi.org/10.4103/jiag.jiag_15_25
20. National Institute for Health and Care Research. Point-of-care devices for detecting diabetic polyneuropathy. Horizon Scan Report 0046. London: NIHR; 2024 [cited 2026 Jan 12]. Available from: https://www.community.healthcare.mic.nihr.ac.uk/reports-and-resources/horizon-scanning-reports/horizon-scan-report-0046
21. Boulton AJ, Armstrong DG, Albert SF, Frykberg RG, Hellman R, Kirkman MS, et al. Comprehensive Foot Examination and Risk Assessment. Diabetes Care. 2008; 31 (8). Available from: https://doi.org/10.2337/dc08-9021
22. Tesfaye S, Boulton AJ, Dyck PJ, Freeman R, Horowitz M, Kempler P, et al. Diabetic Neuropathies: Update on Definitions, Diagnostic Criteria, Estimation of Severity, and Treatments. Diabetes Care. 2010; 33 (10). Available from: https://doi.org/10.2337/dc10-1303
23. England JD, Gronseth GS, Franklin G, Carter GT, Kinsella LJ, Cohen JA, et al. Practice Parameter: Evaluation of distal symmetric polyneuropathy: Role of autonomic testing, nerve biopsy, and skin biopsy (an evidence-based review) [RETIRED]. Neurology. 2009; 72 (2). Available from: https://doi.org/10.1212/01.wnl.0000336345.70511.0f
Copyright
©2026 (Vetriselvan ) et al. This is an open-access journal, and articles are distributed under the terms of the Creative Commons Attribution License CC-BY 4.0. (https://creativecommons.org/licenses/by/4.0/) which permits unrestricted use, distribution, and reproduction in any medium, provided the original authors and source are credited.
Cite this article
Vetriselvan T, Nethaji P, Giriappa D. Prevalence of Diabetic Peripheral Neuropathy among Diabetes Mellitus Patients in a Selected Tertiary Care Center in South India, using Point-of-Care Technology - An Analytical Cross-Sectional Study. Perspectives in Medical Research 2026; 14(1):15-21 DOI: 10.47799/pimr.1401.25.82